Adding an AccuVap Concentration Module to the PrepLinc System eliminates the need for a separate bulk evaporation step when combined with processes like SPE or GPC Cleanup. AccuVap offers concentration of solvent directly from another process (GPC or SPE), AccuVap FLX also allows offline concentration.
Sample concentration with solvent exchange
Unlike other semi-automated evaporation systems, the AccuVap™ will automatically solvent exchange and quantitatively transfer your sample to a GC vial ready for analysis. Powerful software controls heat and vacuum at every stage of the process to protect analyte recoveries. Programmable heated rinses eliminate carryover.
Save time and improve results. Free up lab technicians and decrease sample handling. Adding the AccuVap™ to your PrepLinc™ system will really automate your world!
AccuVap Module
Standout Features:
- Enclosed evaporation chamber
- Three heating zones with user defined heat & vacuum for each zone
- Vacuum control from 100-760 torr
- Two endpoint settings: dryness or adjustable endpoint (1-5 mL)
- Manage use of up to three solvents for sample exchange, final solvent and rinsing
- Add a pre-evaporation spike
- Add a keeper solution
- Perform multiple solvent exchanges
- Heated rinse cycle with choice of solvent ensures no carry-over
- Transfer concentrated sample into GC vial or other storage vial
AccuVap™ Method Editor
- The PrepLinc™ AccuVap™ Method Editor is full-featured, yet intuitive.
- Parameters for each stage of the concentration process are clearly separated.
- Solvents are programmed at software setup for easy recall when programming methods.
- Heating rates and vacuum settings are adjustable on the fly for quick method development.
- Knowledgeable applications staff are available to optimize method parameters for matrix, solvent and analytes of interest.
AccuVap Options
Inline or FLX Model
Choose your AccuVap model based on intended uses. Consult the lists below to determine the model best suited for your lab.
AccuVap™ Inline
- GPC with Evaporation
- SPE with Evaporation
AccuVap™ FLX
- GPC with Evaporation
- SPE with Evaporation
- Evaporation Only
Optional PL GPC Cleanup Module
Optional PL SPEi Starter Kit
(can add 4 more column modules)
Solvent Recovery Module & Chiller
Collects and condenses solvent vapor from concentration process for disposal or recycling
SRM Condenser Module
Condenses solvent vapor and drains to vessel for disposal or recycling.
Collects up to 95% of solvent vapors
Immersion Chiller
Chiller options for SRM
module.
140 W at -30°C
Evaporation Chamber Options
Choose an evaporation chamber based on the desired final volume.
AV081 - Standard
For final volumes from 1 mL to 10 mL
AV082 - 5mL Tip
For final volumes from 0.5 mL to 5 mL
AV083 - 10 mL Tip
For final volumes from 0.5 mL to 5 mL
For small volume and single addition
concentrations
PrepLinc Module Options
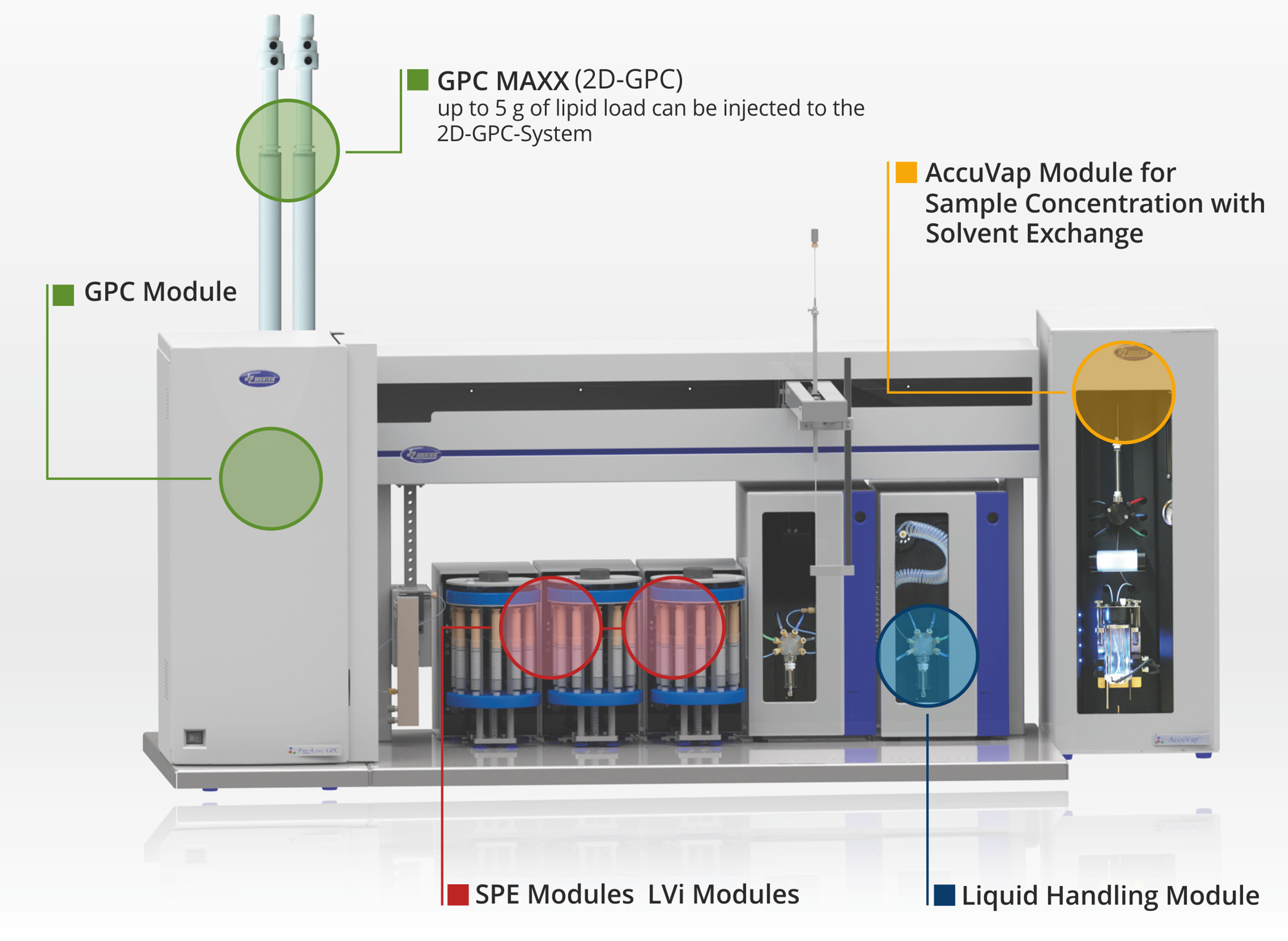
add PrepLinc SPEi Cartridge Modules
- Add two column modules with AS2 autosampler
- Add five column modules with AS4 autosampler
add GPC Direct Inject or Loop Overfill System
- automated GPC cleanup of a wide range of sample matrices
- upgrade to GPC MAXX for increasing the lipid capacity
add PrepLinc GPC MAXX
- greatly increasing the lipid capacity of your GPC system
- upgrade available for any PrepLinc system
add AccuVap Inline or FLX Modules
- Inline offers GPC + Evap mode
- FLX offers GPC + Evap mode and Evap Only mode
add LVi Modules for Water Extraction
- LVi cartridge module with pump modules available
- for large volume samples from 20 ml > 1 liter
Downloads
Application Notes
Application Note 105
Gel Permeation Chromatography is a size-exclusion liquid chromatography method used to remove lipids, sulfur and other co-extractives from environmental and food matrices prior to analytical analysis. It is a desirable technique because it is non-destructive and separates based on molecular size.
GPC Cleanup, while a beneficial cleanup technique, has been criticized for its solvent and time investments. GPC Cleanup using the traditional glass column requires one hour per sample and about 300mL of mobile phase solvent. Additionally, the traditional mobile phase is methylene chloride (DCM), a chlorinated solvent that requires expensive disposal.
To significantly decrease the cost of GPC Cleanup, the run time must be shortened. Simply increasing the mobile phase flow rate may speed the processing time, but will not decrease the amount of solvent used. It will also create pressure problems with the column. Another way to increase sample throughput while decreasing solvent consumption is to decrease the bed volume of the column. The lower bed volume will decrease the run time, thus decreasing the amount of solvent used to process each sample. There are, however, drawbacks to decreasing column bed length in some situations.
Please request your copy at J2 Scientific GmbH.
Application Note 110
The USDA FSIS National Residue Program mandates the testing of domestic meat to prevent violative levels of persistent pollutants like chlorinated pesticides from entering the food supply. Recent findings have prompted an interest in flame retardant levels in meat. Flame retardant compounds, like hexabromobiphenyl, are commonly found in flame retardants and enter the animal by ingestion of retardant-treated items. Little is known of the toxicity of fire retardant compounds in humans, but research in rodents suggests they are associated with cancer, endocrine disruption and brain impairment. Like chlorinated pesticides, fire retardant compounds are highly lipophilic and tend to accumulate in fatty tissue of animals in the food chain.
The standard method for determining chlorinated pesticide residue levels in meat employs GPC Cleanup with GC/ECD detection. In this study the flame retardant compound hexabromobiphenyl was simultaneously determined with a standard list of 20 chlorinated pesticides. Advances in GPC Cleanup column technology allows for a decrease in run time, keeping the entire procedure, extraction through analysis, close to 1 hour per sample.
Please request your copy at J2 Scientific GmbH.
Application Note 132
Currently, 12 substances are regulated by the Stockholm Convention on Persistent Organic Pollutants (POPs) signed in May 2001 by 127 countries, and the work on finding new candidate chemicals to the convention has started. One group of substances in focus is polycyclic aromatic hydrocarbons (PAHs). They are formed during all types of incomplete combustion of organic matter, and they exhibit the characteristic POPs properties: persistence, bio-accumulation, adverse effects and potential for long-range environmental transportation to a certain extent. Many of the PAHs are carcinogenic, they are also believed to exhibit reproductive effects, as well as immune system inhibiting properties, genotoxicity and mutagenicity.
The development of innovative analytical methods for determination of PAHs has been and is of fundamental importance, due to the high carcinogenicity of these compounds. The quali-quantitative analysis of PAHs is an important challenge due to the low concentration at which these hydrocarbons may be present.
Please request your copy at J2 Scientific GmbH.
Application Note 131
The use of Dietary Supplements by consumers has grown from <10% of the population to ~50% of the population over 10 years (US). Ginseng, one of the most popular botanical supplements, is a root crop requiring 4-7 years to mature. The long growing period increase the risk of fungal and insect attack. Numerous chlorinated pesticides, namely PCNB (Quintozene) and Tricyclazole are frequently found in ginseng samples. FDA & private laboratory testing revealed major contamination problems in the 1990’s which still persist today making residue monitoring a high priority. While many supplements can be routinely tested using modern techniques (QuEChERS), many chlorinated residues and high-lipid/saponin matrices can create challenges for this modern technique. Florisil column clean up (SPE) and Gel Permeation Chromatography (GPC) are utilized for such matrices. Historical GPC involved significant time and resources from the analyst to collect fractions, evaporate them and perform various solvent exchanges & SPE. Using the new-generation PrepLinc™ system, these functions are fully automated creating a ready-to-inject sample in an autosampler vial making GPC viable again for production laboratories. This poster (the first in a series) will introduce the system and briefly outline the methods utilized for sample preparation & provide matrix examples. Future posters/papers will provide further detail on the project.
Please request your copy at J2 Scientific GmbH.
Application Note 120
Extract purification for pico- or nanogram scale GC/MS is time consuming, laborious and costly, and may suffer from performance variations in manual cleanup chromatography. Size exclusion (GPC) followed by adsorptive chromatography is useful for cleanup of biota, soil and sediment extracts for high resolution PCDD/PCDF analyses. GPC cleanup of extracts is allowed or encouraged in several EPA methods and is of great value to laboratories practicing such analyses. Sample extract cleanup for PCDD./PCDF always involve a carbon column stage. Sample concentrate, typically after other cleanups, is passed onto carbon and forward elution drives out various interferences. Reverse elution utilizes solvent containing a component (e.g. toluene) having great affinity for carbon. This disgorges PCDD/PCDF congeners into an eluate ready for evaporation and analysis.
In this study GPC eluate was forward eluted through a carbon cartridge (bed of powder mixed with granular substrate, packed between two frits) placed in line after the GPC column. During forward elution, target compounds collect at or near the column head while interferences are flushed forward. Valve switching enables reverse elution with toluene for collection of targets. Thus PCDD/PCDF sample cleanup can be conducted in highly automated fashion with minimal operator contact.
Please request your copy at J2 Scientific GmbH.
Application Note 30
Chlorination of drinking water has been a commonly practiced method of disinfection for over a century. However, the disinfection by-products arising from the reaction of organic material in the water and chlorine may give rise to certain aberrant carcinogenic effects. EPA Method 552.1 and 552.3 prescribe procedures for testing of haloacid constituents in drinking water by ion-exchange or micro extraction, followed by esterification and quantitation by GC-ECD.
In this study, the 552.1 ion-exchange method is automated for different water samples to afford increased reproducibility, unattended operation, and consistency of sample loading and elution.
Please request your copy at J2 Scientific GmbH.
- Maximizing Lipid Load With 2-Dimensional GPC Cleanup
- QuEChERS, SPE and GPC: A Comparison of Sample Preparation Techniques for Analysis of Pesticides in Problematic Matrices
- Automated GPC with Inline SPE to Improve Sample Cleanup Without Adding Time or Solvent
- Additional cleanup for DIN EN 12393 minimising matrix effects and improving result quality in GC-MS
- A Combined SPE Method for Analysis of Chloroacetic Acids in Drinking Water
- Bestimmung von leichten und schweren polycyclischen aromatischen Kohlenwasserstoffen (PAKs) in Fetten und Ölen pflanzlicher und tierischer Herkunft mittels automatisierter 2D-GPC und anschließender GC-MS-Detektion
- Automatisierte Probenvorbereitung für die Bestimmung von Pestizidrückständen in hocheffizienten Laboratorien mit GPC-GC-MS/MS und -LC-MS/MS
- Maximierte Probenaufgabe mit zweidimensionaler Gelpermeationschromatografie (2D-GPC)
- Automatisierung der Bausteine GPC, C1 und C2 der Methode L 00.00-34
- Cleanup-Methode für Nahrungsergänzungsmittel wie z.B. Ginseng
- Modifizierte Cleanup-Methode für Dioxine und persistente organische Schadstoffe (POPs)
- Automatisierung der Wasserextraktion mit dem SPE-Wasserextraktionssystem LVi
- Traditionelles Dioxin-Cleanup mit dem PrepLinc System
- Wiederauffindungsraten unterschiedlicher Pflanzenschutzmittel mit GPC und AccuVap
- Erfahrungen zur Automatisierung des GPC-Reinigungsver fahrens bei der Untersuchung von tierischen Lebensmitteln auf Chlorkohlenwasserstoffe
- Der Einsatz der automatischen Gelchromatographie zur Reinigung von Pesticidextrakten Organochlor
- Pflanzenbehandlungsmittel in Tabak und Tabakerzeugnissen
- Bestimmung der Fungizide Bitertanol, Fuberidazol, Imazalil, Rabenzazole, Triadimefon und Triadimenol in Pflanzen und Boden
- Methode zur Aufarbeitung von Lebensmitteln und Futtermitteln pflanzlicher und tierischer Herkunft für die Multirückstandsbestimmung lipoid- und wasserlöslicher Pflanzenbehandlungsmittel
- Zur Analytik von Chlorkohlenwasserstoffen in Zwiebeln nach Reinigung mit der Gelpermeationschromatographie
- Schnelle Untersuchung von Milch auf chlorierte Kohlenwasserstoffe mittels automatischer Gelchromatographie
- Automatisierte Gelchromatographie als Reinigungsverfahren zum Nachweis von ECD-erfaßbaren Wirkstoffen, chlorierten Kohlenwasserstoffen, Pentachlorphenol sowie von Diphenyl und o-Phenylphenol in pflanzlichen Materialien
- Untersuchungen zum Einsatz der Gelpermeationschromatograpie in der Rückstandsanalytik
- Nachweis von Aflatoxin B1 in Futtermitteln für Milchtiere
- Bestimmung der Rückstände von aromatischen Dinitroverbindungen mittels gelchromatischer Reinigung
- Die Gelpermeationschromatographie, eine universelle Reinigungsmethode in der Analytik von Pflanzenschutzmitteln
- Untersuchungen zur Messung und Bewertung von Rückständen des Ektoparasitenbekämpfungsmittels Phoxim in Milch
- Methode zur Aufarbeitung von Lebensmitteln und Futtermitteln pflanzlicher und tierischer Herkunft für die
- Multirückstandsbestimmung lipoid- und wasserlöslicher Pflanzenbehandlungsmittel
- Untersuchungen zur Gelchromatograpie (GPC) als Reinigungsverfahren in der Rückstandsanalytik von Tierarzneimitteln
- Eine schnelle Methode zur Bestimmung des Ebergeruchsteroids Androstenon
- Analysenverfahren zur Bestimmung von polychlorierten Dibenzodioxinen und Dibenzofuranen in Frauenmilch
- Untersuchungen zum Vorkommen von Moschus-Xylol in Fischen
- GPC-clean up von fetthaltigen Matrizes in der Rückstandsanalytik unter Verwendung von OPTIMA-Säulen
- Entwicklung einer Methode zur Bestimmung von Nitromoschusverbindungen im Hausstaub
Contact
J2 Scientific GmbH
Reifenstuelstraße 3
80469 Munich / Germany
Tel.: +49 (0) 89 72069587
E-Mail: info@j2scientific.eu
Distributor
ANTEC GmbH
Analysen- und Prozesstechnik
Hauptstraße 4
82404 Sindelsdorf
Tel.: +49 (0) 8856 9910




